Pilots and air traffic controllers to be monitored for 48 hours after taking coronavirus vaccine “to ensure aviation safety”
01/28/2021 / By Zoey Sky
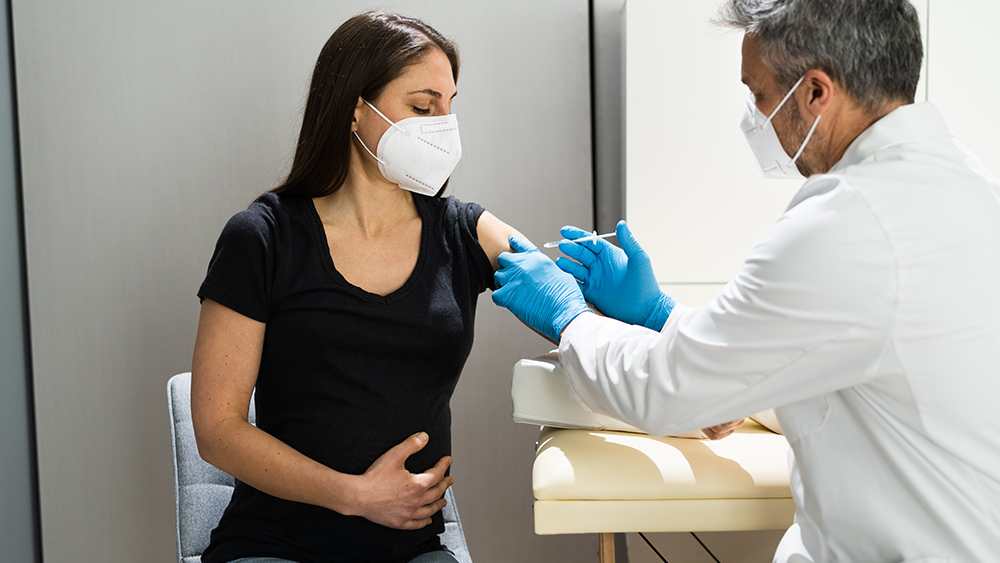
Although the Food and Drug Administration (FDA) has deemed the coronavirus vaccine safe for use, the Federal Aviation Administration (FAA) chooses to err on the side of caution.
The FAA authorized the use of the Moderna COVID-19 vaccine for pilots and air traffic controllers last Dec. 19. However, the FAA required a 48-hour wait period before pilots and controllers could begin “conducting safety-sensitive aviation duties,” similar to what the agency did with the Pfizer-BioNTech vaccine.
The policy was released a day after the FDA granted emergency use authorization for mRNA-1273 or the Moderna vaccine.
Pfizer-BioNTech’s vaccine received FDA authorization on Dec. 11 and the FAA permitted its use on Dec. 12.
The FAA Office of Aerospace Medicine said: “Holders of FAA-issued Airman Medical Certificates or Medical Clearances may receive the Pfizer-BioNTech or Moderna COVID-19 vaccine. However, a 48-hour no fly/no [safety-related] duty interval must be observed after each dose.”
The FAA advised inoculated pilots and air traffic controllers that they are forbidden from performing flight crewmember duties or air traffic control duties if they don’t “meet medical certification requirements, including those related to adverse events from medications that render them unable to perform such duties.”
While the FDA authorizes additional vaccines, the FAA evaluates and releases policies. The FAA emphasized that it will strictly observe the patient response to both Pfizer-BioNTech and Moderna vaccines. The agency added that it may adjust the policy as needed “to ensure aviation safety.”
Air traffic controllers and pilots helped ship the first doses of the Pfizer-BioNTech vaccine last Dec. 13. Both Houston Mills, who is an Aircraft Owners and Pilots Association (AOPA) member and UPS Inc. Vice President of Flight Operations and Safety, and first officer Neal Newell safely flew the cargo from Lansing, Michigan, to Louisville, Kentucky.
AOPA President Mark Baker praised pilots and air traffic controllers who are at the forefront of the fight against the ongoing COVID-19 pandemic as they delivered personal protective equipment and the vaccines.
“I am pleased the FAA is acting quickly to evaluate and allow pilots and controllers to take either of the FDA-approved vaccines and return to work and flying after a short period,” concluded Baker.
Back in December, the FAA sent guidance to airports to prepare for vaccine distribution. The agency also advised other facilities that may serve as alternate or diversion airports.
Additionally, the FAA announced that some aircraft used for vaccine shipments may be larger than the aircraft used for passenger-carrying flights that usually serve that airport. Operators were advised to have extra aircraft rescue and fire-fighting services.
Are experimental vaccines really “safe?”
Many believe the coronavirus vaccines are still experimental and, like other experimental drugs, are linked to negative side effects.
For example, a CBS 60 Minutes’ segment called “The Swine Flu Fraud of ‘76” revealed shocking details about a vaccine that injured thousands of Americans. Those who received the vaccine even sued the U.S. government. (Related: Woman suffers whole body convulsions after taking experimental Moderna COVID-19 vaccine.)
The FDA reported that Moderna’s Covid-19 vaccine causes common side effects such as fatigue, headaches and muscle pain. Other rare symptoms of the vaccine include intractable nausea (vomiting that is difficult to control) and facial swelling potentially triggered by the shots.
Additionally, over nine in 10 participants who were vaccinated felt pain at the injection site. At least seven volunteers in 10 felt fatigued and at least six out of 10 had headaches or muscle pain.
Over 44 percent of people who were vaccinated experienced joint pain and more than 43 percent experienced chills. The agency also found that more severe side effects were recorded in 0.2 percent to 9.7 percent of volunteers. These serious symptoms were more common after they received the second dose.
Moderna’s vaccine requires two shots that are given within several weeks. The FDA reported that at least 15 percent of vaccinated participants got a fever after receiving either the first or second dose.
Less than six percent reported symptoms that lasted for a week or so after vaccination. According to the agency, some of the trial participants had a fever that lasted more than a week.
While there were seven “serious adverse events” in the COVID-19 vaccine trial, the FDA insisted that none of them was fatal. Four were attributed to the vaccine by trial investigators and Moderna, with side effects including intractable nausea and vomiting, facial swelling and rheumatoid arthritis.
While the Moderna coronavirus vaccine is supposedly safe to use, the FDA recommends monitoring those who were vaccinated to see if anyone develops Bell’s palsy. Out of the trial’s 30,000 participants, four individuals experienced the condition that causes half of the face to droop.
Visit VaccineInjurynews.com to read more articles about the potential side effects of the coronavirus vaccine.
Sources include:
Tagged Under: adverse events, Big Pharma, BioNTech, BNT162b2, covid-19, immunization, pandemic, Pfizer, public safety, side effects, vaccine injury, vaccine reactions, vaccines, Wuhan coronavirus
RECENT NEWS & ARTICLES
VaccineDamage.News is a fact-based public education website published by Vaccine Damage News Features, LLC.
All content copyright © 2018 by Vaccine Damage News Features, LLC.
Contact Us with Tips or Corrections
All trademarks, registered trademarks and servicemarks mentioned on this site are the property of their respective owners.